| Admissions | Contact Us | Examinations | Grants | Instructors | Lecture | Membership | Students login | Schools | Colleges | Universities | Professional Examinations | Recommendations | Research Grants | Search | Librarians | Forms | Booksellers | Continents/States/Districts | Contracts | Volunteer |
Annotation or definition
|
What is a Battery?
Batteries are devices that convert stored chemical energy into useful electrical energy. A battery may be thought of as a clever variant of a standard exothermic chemical reactor that yields chemical products with lower energy content than the chemical reactants. In such a chemical reactor, the overall chemical reaction proceeds spontaneously (possibly requiring a catalyst and/or elevated temperature) when the reactants are brought into physical contact. In a battery, the overall chemical reaction is divided into two physically and electrically separated processes: one is an oxidation process at the battery negative electrode wherein the valence of at least one species becomes more positive, and the other is a reduction process at the battery positive electrode wherein the valence of at least one species becomes more negative. The battery functions by providing separate pathways for electrons and ions to move between the site of oxidation and the site of reduction. The electrons pass through the external circuit where they can provide useful work, for example power a portable device such as a cellular phone or an electric vehicle. The ions pass though the ionically conducting and electronically insulating electrolyte that lies between the two electrodes inside the battery. Therefore, the ionic current is separated from the electronic current, which can be easily controlled by a switch or a load in the external circuit. When a battery is discharged, an electrochemical oxidation reaction proceeds at the negative electrode and passes electrons into the external circuit, and a simultaneous electrochemical reduction reaction proceeds at the positive electrode and accepts electrons from the external circuit, thereby completing the electrical circuit. The change from electronic current to ionic current occurs at the electrode/electrolyte interface. Faraday’s Law, which describes the quantitative proportional relationship between the equivalent quantities of chemical reactants and electrical charge, governs this change. When one attempts to recharge a battery by reversing the direction of electronic current flow, an electrochemical reduction reaction will proceed at the negative electrode, and an electrochemical oxidation reaction will proceed at the positive electrode. The common non-rechargeable (throwaway) alkaline battery uses a zinc negative electrode, a manganese dioxide positive electrode, and an aqueous alkaline electrolyte. The overall battery cell discharge reaction is Zn + MnO2 + H2O = ZnO + Mn(OH)2. For this chemical reaction the free energy of the reaction products is lower than the free energy of the reactants, and therefore the reaction proceeds spontaneously. During battery cell discharge the electrochemical oxidation reaction at the negative electrode is Zn + 2OH� = ZnO + H2O + 2e�, and the electrochemical reduction reaction at the positive electrode is MnO2 + 2e� + 2H2O = Mn(OH)2 + 2OH�. One can use the well-known thermodynamic free energy change of the chemical reaction Zn + MnO2 + H2O = ZnO + Mn(OH)2 along with the Faraday Law to calculate the observed voltage of this cell, which is 1.5 volts. The modern rechargeable lithium-ion battery uses a lithium-carbon negative electrode, a cobalt dioxide positive, and an electrolyte that contains a lithium salt dissolved in an organic solvent. The overall battery discharge reaction is LiC6 + CoO2 = C6 + LiCoO2. As for the throwaway alkaline battery, the free energy of these reaction products is lower than the free energy of the reactants, and therefore the reaction proceeds spontaneously. During battery cell discharge, the electrochemical oxidation reaction at the negative electrode is LiC6 = Li+ + C6 + e�, and the electrochemical reduction reaction at the positive electrode is CoO2 + Li+ + e- = LiCoO2. One can use well-known thermodynamic free energy change of the chemical reaction LiC6 + CoO2 = C6 + LiCoO2 along with the Faraday Law to calculate the observed voltage of this cell, which is 4.0 volts. In the case of the rechargeable battery, the electrochemical oxidation-reduction reactions are reversible at both electrodes. For example, when the battery is recharged, the overall electrochemical reduction reaction at the negative electrode is identical to the electrochemical oxidation reaction that proceeded at the negative electrode when the battery was discharged, only written in reverse. In the case of the non-rechargeable battery, when one attempts to recharge the battery by reversing the direction of electron current flow, at least one of the electrochemical oxidation-reduction reactions is not reversible. For example, when the battery is charged, the overall electrochemical reduction reaction that proceeds at the negative electrode may not be the reverse of the electrochemical oxidation reaction that proceeded when the battery was discharged. For example, metal oxidation might be the sole oxidation reaction during battery discharge, whereas gas formation might be a significant reduction reaction during battery recharge (a potentially dangerous condition!). An added requirement for a well-behaved (i.e., long-lived and safe) rechargeable battery is that not only must the electrochemical oxidation-reduction reactions be reversible; they must also return the electrode materials to their original physical state. For example, if rough or filamentary structures are formed after repeated charge-discharge cycles, then there may result unwanted electrode growth and subsequent electronic contact between the battery electrodes, i.e. an internal short circuit that rapidly discharges the battery. Because of these requirements, the development of a well-behaved rechargeable battery is significantly more difficult than the development of a non-rechargeable battery. As described above, the three fundamental components of a battery are the negative electrode (usually identified as the anode, where anodic oxidation proceeds when the battery is discharged); the positive electrode (usually identified as the cathode, where cathodic reduction proceeds as the battery is discharged); and the electrolyte (which is ionically conducting and electronically insulating). Now, let us examine in more detail each of these three battery components. We will focus on the components for the rechargeable lithium-ion battery because it and related batteries with non-aqueous electrolytes are considered to be the most promising technology for powering electric vehicles, hybrid-electric vehicles, and plug-in hybrid vehicles. The anode must have a low mass per unit of charge delivered so that a high-energy battery can be developed, support high oxidation and reduction rates to allow rapid battery charge and discharge, exhibit a low electrochemical potential so that a high-voltage battery can be developed, show high coulombic (current) efficiency so that unwanted side reactions do not occur, be environmentally benign and safe, exhibit very long lifetime with little performance decay, and be low cost. To support high reaction rates that are suitable for vehicle applications, the anode must have a high surface area, and therefore anodes are typically fabricated using LiC6 carbons in the form of small particles. Also, the anodes must be designed to provide intimate contact between the electrolyte, the LiC6 carbon, and a so-called current collector (typically a thin copper sheet) so that electrons and ions have ready access to the sites of electrochemical reactions. This requirement usually leads to the addition of an extra carbon phase to enhance electronic conduction. Finally, the anode must not fall apart as the battery is charged and discharged, therefore a polymeric binder is used to hold the anode together. All of these requirements are very demanding and often conflict with one another. So, it is a very difficult task to arrive at an optimal set of materials and a suitable design for a modern, high-performance lithium-ion battery anode. The BATT Program conducts sophisticated fundamental and focused anode research to do exactly this, as well as explore the use of alternative anode materials such as lithium alloys. Similarly, the cathode must have a low mass per unit of charge delivered so that a high-energy battery can be developed, support high oxidation and reduction rates to allow rapid battery charge and discharge, exhibit a high electrochemical potential so that a high-voltage battery can be developed, show high coulombic (current) efficiency so that unwanted side reactions do not occur, be environmentally benign and safe, exhibit very long lifetime with little performance decay, and be low cost. The requirements for the cathode parallel those of the anode, as described above. In addition, because the most promising cathode materials are brittle ceramics composed of metal oxides, they are susceptible to cracking and other forms of physical degradation as lithium ions are inserted and extracted. The BATT Program conducts leading cathode research to address these difficult problems. The electrolyte must have high ionic conductivity to allow the development of a high-power battery, be stable at both the high potential of the battery cathode and the low potential of the battery anode, be compatible with a physical separator (usually a porous polymeric material) that prevents physical or electronic contact between the anode and cathode, adequately wet the anode, cathode, and separator, be environmentally benign, have low vapor pressure, and be low cost. |
Aircraft Batteries

Aviation Requirements Batteries used for aviation applications may be of either the primary (single use) type or the secondary (rechargeable) type. Any battery intended for use as a power source for equipment installed or routinely carried on aircraft must not only be safe but ideally have a high energy density, be lightweight, require minimal maintenance, reliable and able to operate efficiently over a wide environmental envelope. Battery manufacturers continue to develop new technologies in an attempt to achieve these ideals but in many cases compromises in these non-safety objectives are necessary and in some cases, safety implications of new designs have been overlooked, particularly in respect of the rapidly increasing use of Lithium batteries. Battery Types Numerous types of batteries have been developed and variants of some of them are used in aviation applications. These include: Lead Acid. A lead acid battery cell contains an anode made from lead oxide and a cathode of elemental lead immersed in an electrolyte solution of sulfuric acid. In some lead acid batteries, the electrolyte is suspended in a silica gel or impregnated into a fiberglass mat to make the battery non-spillable. While lead acid batteries have good energy storage and power provision properties, they are quite heavy and their energy density is relatively low. If overcharged, lead acid batteries can sometimes vent hydrogen gas which can result in an explosion or lead to a fire. Lead acid batteries are often used as the main battery(s) in an aircraft. Nickel Cadmium (NiCd). Nickel-cadmium cells have an anode made of cadmium hydroxide and a cathode of nickel hydroxide that are immersed in an electrolyte made up of potassium, sodium and lithium hydroxides. Nickel-cadmium batteries require relatively low maintenance, are reliable and have a wide operating temperature range. NiCd batteries are subject to memory effect and may experience thermal runaway if overcharged. Many countries impose strict disposal regulations on NiCd batteries because of the heavy metals used in their manufacture. NiCd batteries are suitable for many aircraft applications inclusive of main aircraft batteries. Nickel-Metal Hydride (Ni-MH). Nickel-metal hydride cells have an anode made of a metal alloy capable of absorbing and releasing hydrogen. The cathode is made from nickel hydroxide and both are immersed in an electrolyte solution of potassium, sodium and lithium hydroxides. Small capacity cells of this type of battery are sealed and are maintenance free. Their principal shortcoming is that they require precise charge level monitoring to control gaseous exchanges and to minimize heating while under charge. Ni-MH batteries have a high energy density and are ideal for high-capacity requirements. In aircraft, Ni-MH batteries are often used to power systems such as the emergency door and floor escape path lighting as well as portable entertainment devices and electronic flight bags. Lithium-ion/Lithium-polymer. Lithium-ion (Li-ion) and Lithium Polymer (Li-poly) batteries, also described as 'lithium secondary batteries' are rechargeable. Their cells have an anode of graphite and a cathode consisting of a combination material which is able to accept and release lithium ions repeatedly (for recharging) and quickly (for high current) such as lithium manganese oxide (Li-Mn2O4). A non-aqueous electrolyte, mainly comprised of a mixture of organic carbonates, is used. Charging or discharging a Li-Ion battery involves an exchange of lithium ions between the electrodes. Typical cell output voltage is between 3 and 4.2 volts depending primarily on the materials used to construct the cathode. Lithium Metal. Lithium Metal batteries, also described as 'lithium primary batteries' are non-rechargeable. Their electrochemistry is most commonly based on Lithium-Manganese Dioxide (Li-MnO2) cells which have a graphite anode and a Lithium dioxide cathode. Threats There are a number of potential threats that can be associated with aircraft batteries, their distribution networks and their charging and monitoring systems. These threats include: Battery Leakage. Overfilling a wet cell battery can cause leakage. Likewise, damage to the battery case caused by mishandling, overcharging or freezing can result in leakage. Battery Internal Failure or Short Circuit. Manufacturing defects or inappropriate handling can result in internal failures. Battery Overcharging. Batteries can be overcharged due to faulty charging equipment or inappropriate maintenance practices. Excessive Battery Charging Rate. Some battery types are vulnerable to high rates of charge. Excessive Battery Discharge Rate. Some battery types are vulnerable to high rates of discharge. Battery Bus Fault or Fire. A Battery Bus Bar is "hot" - it cannot be electrically isolated from the source battery without physically removing the battery. Effects The effects which could result from the threats, as listed above, range from minor to potentially catastrophic depending upon the circumstances of the occurrence and the type of battery involved. As examples: leakage from a spillable lead acid battery could result in corrosion, component damage or injury. overcharging of a lead acid battery could result in an explosion. Overcharging, excessive charge rate or excessive discharge rate in a lithium-ion battery could result in a thermal runaway leading to battery explosion or fire. This, in turn, could lead to injury or death and collateral damage up to the potential loss of the aircraft. while technically not a battery fault, a problem on an associated "hot" battery bus bar could result in fumes, smoke or fire. Defences Mitigation of most of the threats associated with aircraft batteries can be achieved through robust design, testing, maintenance and operational practices and procedures. These include: Aircraft Design. Procedures for the Certification of aircraft and their equipment should involve validation of their safety when malfunctioning such as battery containment commensurate with internal pressure and/or venting of hazardous fumes and substances which may occur. New Technologies. Certification must pay particular attention to new technologies, especially where their development or transfer to aircraft and their equipment is rapid. Maintenance Practices. Should follow the regulatory requirements and manufacturers' guidance in respect of inspection, recharging, removal and replacement criteria. Flight Crew Procedures. Should take full account of manufacturers' direction on normal, abnormal and emergency system usage and monitoring. AOM/QRH Guidance. Should provide clear, unambiguous information on system limitations and on the actions to be followed in the event of an exceedance or malfunction. Terms There are numerous terms used to describe batteries, their component parts and specific battery related conditions, problems or issues. These include: Anode. An anode is the positive electrode of a voltaic cell. It is the electrode at which an oxydation reaction takes place resulting in the loss of electrons. Cathode. A cathode is the negative electrode of a voltaic cell. It is the electrode at which a reduction reaction takes place resulting in the gain of electrons. Dry Cell. A dry cell battery is a "non-spillable" type battery in which the electrolyte is immobilized in a gel or a paste. Electrolyte. A chemical compound which, when fused or dissolved in certain solvents, usually water, will conduct an electric current. All electrolytes in the fused state or in solution give rise to ions which conduct the electric current. Energy Density. Energy density is the amount of energy that can be stored per unit volume. Memory Effect. Memory Effect is a phenomenon in which a cell, if discharged in successive cycles to a point that is less than its full depth of discharge, will temporarily lose the remainder of its capacity. Parallel Connection. The arrangement of cells in a battery made by connecting all positive terminals together and all negative terminals together. The total voltage of the group of cells is the same as that of one cell and the current drain is divided equally among the cells within the group. Primary Battery. A primary battery is one that produces current as soon as its components are assembled. It is considered a "disposable" battery as it is not rechargeable and therefore has a limited useful life. Secondary Battery. A secondary battery is rechargeable. In most cases, it must be charged before first use as it is normally assembled with the active components in a discharged state. A secondary battery is both initially charged and recharged by applying an electric current which reverses the chemical reactions that take place during normal battery useage. The required electrical current is applied in a controlled fashion by using a battery charger. Series Connection. The arrangement of cells in a battery made by connecting the positive terminal of each successive cell to the negative terminal of the next adjacent cell so that their voltages are cumulative. Thermal Runaway. Thermal runaway is a situation that occurs when an increase in temperature changes the conditions of a reaction in a way that causes a further increase in temperature. In other words, if a process is accelerated by an increase in temperature and that acceleration results in the release of additional energy which further increases the temperature, a state of thermal runaway is said to exist. This state can lead to an explosion, fire or other destru-ctive result. Voltaic Cell. A voltaic (or galvanic) cell is an electrochemical cell that derives electrical energy from a spontaneous redox reaction taking place within the cell. Wet Cell. A wet cell battery is one containing an electrolyte in liquid form. These are sometimes referred to as "spillable" batteries. Aircraft Electrical Systems 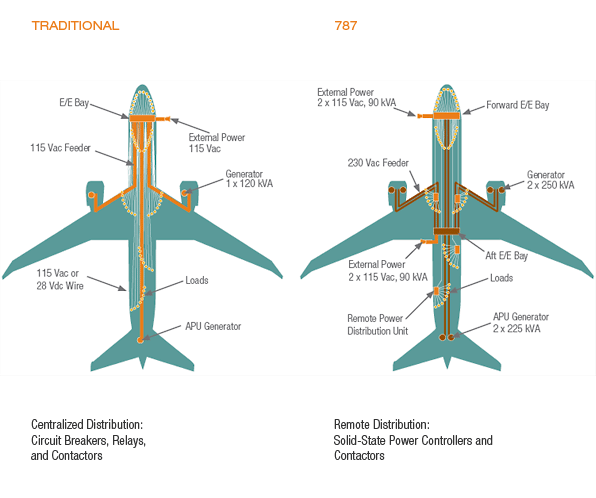
The satisfactory performance of any modern aircraft depends to a very great degree on the continuing reliability of electrical systems and subsystems. Improperly or carelessly installed or maintained wiring can be a source of both immediate and potential danger. Here are further guidelines. |
Automobile Battery
Car Battery
Bike Battery
E-Bike Battery
| An automotive battery is a type of rechargeable battery that supplies electric energy to an automobile. Automotive SLI batteries are usually lead-acid type, and are made of six galvanic cells in series to provide a 12 volt system. Each cell provides 2.1 volts for a total of 12.6 volt at full charge. Heavy vehicles such as highway trucks or tractors, often equipped with diesel engines, may have two batteries in series for a 24 volt system, or may have parallel strings of batteries. |
Batteries and Advanced Airplanes
|
Electricity has been used in powered flight since the pioneering days of aviation. Orville and Wilbur Wright used an electrical spark to ignite the fuel mixture in the engine that powered the Wright Flyer off the ground and into the history books. Today�s jet airplanes have much more demanding requirements and consequently more advanced electrical systems, of which batteries are an integral component. The 787 Dreamliner has two primary rechargeable batteries � the main and auxiliary power unit (APU). While identical part numbers, they serve separate purposes. The main battery �powers up� aircraft systems, bringing the airplane to life before the engines have been started. Once the engines are started, the electrical energy to run the systems comes from generators. It also is used to support ground operations such as refueling and powering the braking system when the airplane is towed. The main battery also provides backup power for critical systems during flight in the extremely unlikely event of a power failure. It is located in the forward electronics equipment (EE) bay, which is under the main cabin floor at the front of the airplane. The APU battery supplies power to start the APU, which in turn can start the airplane engines. The APU, and its battery, also serves as part of the multiple layers of redundancy that would ensure power in the rare possibility of a loss of primary sources of power. 

Matching the right battery to the requirements After extensive testing, Boeing ultimately selected the lithium-ion type battery because it has the right functionality and chemistry to deliver a large amount of power in a short period of time to do a high-energy task like start a jet engine. It then has the ability to recharge in a relatively short period of time so that it is available for the critical backup role that it plays during flight. Earlier commercial airplane models, such as the 777, 747 and MD-11, used nickel cadmium (NiCd) batteries, which are heavier, larger and less powerful. Batteries, like other technologies, have advanced significantly, and lithium-ion type batteries match up with the unique requirements of advanced aircraft. Lithium-ion batteries have other key advantages that suit it for modern jet application: The required high voltage and high current production Improved power quality An ability to recharge quickly Similar functionality to that of NiCd batteries while weighing 30 percent less Compact � about the size of the average car battery Since entering service, Boeing 787 lithium-ion batteries, each with eight cells, have logged more than 2.2 million cell-hours on the ground and in the air during more than 50,000 flight-hours. No battery-related incidents occurred before January 2013, when the airplane experienced two events. Investigation into these events is in progress. Boeing has been using lithium batteries for decades safely and successfully in other demanding aerospace applications. For instance, they have been successfully used in the satellite industry. Closer to home, lithium batteries are being used safely to power everything from consumer electronics, household power tools and many other applications. |
Battery History
History of Battery Development
|
Timeline of Battery History
1748 - Benjamin Franklin first coined the term "battery" to describe an array of charged glass plates. 1780 to 1786 - Luigi Galvani demonstrated what we now understand to be the electrical basis of nerve impulses and provided the cornerstone of research for later inventors like Volta to create batteries. 1800 Volta (Italy) Invention of the voltaic cell 1802 Cruickshank (England) First electric battery capable of mass production 1820 Amp�re (France) Electricity through magnetism 1833 Faraday (England) Announcement of Faraday�s Law 1836 Daniell (England) Invention of the Daniell cell 1839 Fuel Cell - William Robert Grove developed the first fuel cell, which produced electrical by combining hydrogen and oxygen. 1839 to 1842 - Inventors created improvements to batteries that used liquid electrodes to produce electricity. Bunsen (1842) and Grove (1839) invented the most successful. 1859 Plant� (France) Invention of the lead acid battery 1859 Rechargeable - French inventor, Gaston Plante developed the first practical storage lead-acid battery that could be recharged (secondary battery). This type of battery is primarily used in cars today. 1868 Leclanch� (France) Invention of the Leclanch� cell 1866 Leclanche Carbon-Zinc Cell - French engineer, Georges Leclanche patented the carbon-zinc wet cell battery called the Leclanche cell. According to The History of Batteries: "George Leclanche's original cell was assembled in a porous pot. The positive electrode consisted of crushed manganese dioxide with a little carbon mixed in. The negative pole was a zinc rod. The cathode was packed into the pot, and a carbon rod was inserted to act as a currency collector. The anode or zinc rod and the pot were then immersed in an ammonium chloride solution. The liquid acted as the electrolyte, readily seeping through the porous cup and making contact with the cathode material. The liquid acted as the electrolyte, readily seeping through the porous cup and making contact with the cathode material." Georges Leclanche then further improved his design by substituting the ammonium chloride paste for liquid electrolyte and invented a method of sealing the battery, inventing the first dry cell, an improved design that was now transportable. 1881 - J.A. Thiebaut patented the first battery with both the negative electrode and porous pot placed in a zinc cup. 1881 - Carl Gassner invented the first commercially successful dry cell battery (zinc-carbon cell). 1888 Gassner (USA) Completion of the dry cell 1899 Jungner (Sweden) Invention of the nickel-cadmium battery 1899 - Waldmar Jungner invented the first nickel-cadmium rechargeable battery. 1901 Alkaline Storage - Thomas Alva Edison invented the alkaline storage battery. Thomas Edison's alkaline cell had iron as the anode material (-) and nickelic oxide as the cathode material (+). 1901 Edison Invention of the nickel-iron battery 1932 Shlecht & Ackermann (Germany) Invention of the sintered pole plate 1947 Neumann (France) Successfully sealing the nickel-cadmium battery 1949 Alkaline-Manganese Battery - Lew Urry developed the small alkaline battery in 1949. The inventor was working for the Eveready Battery Co. at their research laboratory in Parma, Ohio. Alkaline batteries last five to eight times as long as zinc-carbon cells, their predecessors. 1954 Solar Cells - Gerald Pearson, Calvin Fuller and Daryl Chapin invented the first solar battery. A solar battery converts the sun's energy to electricity. In 1954, Gerald Pearson, Calvin Fuller and Daryl Chapin invented the first solar battery. The inventors created an array of several strips of silicon (each about the size of a razorblade), placed them in sunlight, captured the free electrons and turned them into electrical current. Bell Laboratories in New York announced the prototype manufacture of a new solar battery. Bell had funded the research. The first public service trial of the Bell Solar Battery began with a telephone carrier system (Americus, Georgia) on October 4 1955. Mid 1960 Union Carbide Development of primary alkaline battery 1964 - Duracell was incorporated. Mid 1970 Development of valve regulated lead acid battery 1990 Commercialization nickel-metal hydride battery 1992 Kordesch (Canada) Commercialization reusable alkaline battery 1999 Commercialization lithium-ion polymer 2001 Anticipated volume production of proton exchange membrane fuel cell |
Coin cells of various diameters and thicknesses.
 
Assorted sizes of button and coin cells, including alkaline and silver oxide chemistries. Four rectangular 9 V batteries are also shown, for size comparison. Enlarge to see the button and coin cell size code markings. |
Battery chemistry
|
The terminal voltage of a battery cell depends on the chemicals and materials used in its construction, and not on its physical size. For example, primary (non-rechargeable) alkaline batteries have a nominal voltage of 1.5 volts. Rechargeable NiCd (nickel cadmium) and NiMH (nickel metal hydride) typically output 1.25 volts per cell. Devices intended for use with primary batteries may not operate properly with these cells, given the reduction in voltage. Dry Leclanche (carbon-zinc), alkaline and lithium batteries are the most common modern types. Mercury batteries had stable cell terminal voltages around 1.35 volts. From the late 1940s until the mid-1990s, mercury batteries were made in many consumer and industrial sizes. They are no longer available since careless disposal can release toxic mercury into the environment. They have been replaced by zinc-air batteries, which also produce 1.35 volts. The full battery designation identifies not only the size, shape and terminal layout of the battery but also the chemistry (and therefore the voltage per cell) and the number of cells in the battery. For example, a CR123 battery is always Li-MnO2 ('lithium') chemistry, in addition to its unique size. |
Classification of Cells or Batteries
|
Electrochemical batteries are classified into 4 broad categories.
A primary cell or battery is one that cannot easily be recharged after one use, and are discarded following discharge. Most primary cells utilize electrolytes that are contained within absorbent material or a separator (i.e. no free or liquid electrolyte), and are thus termed dry cells. A secondary cell or battery is one that can be electrically recharged after use to their original pre-discharge condition, by passing current through the circuit in the opposite direction to the current during discharge. The following graphic evidences the recharging process. 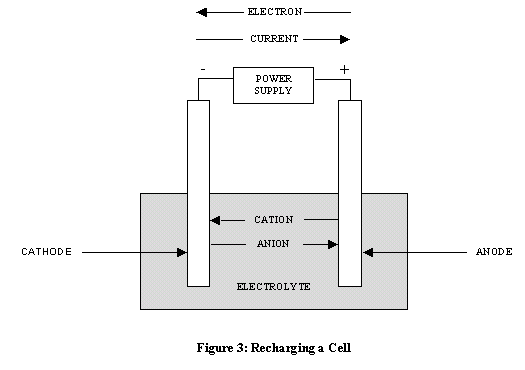 Secondary batteries fall into two sub-categories depending on their intended applications. Cells that are utilized as energy storage devices, delivering energy on demand. Such cells are typically connected to primary power sources so as to be fully charged on demand. Examples of these type of secondary cells include emergency no-fail and standby power sources, aircraft systems and stationary energy storage systems for load-leveling. Cells that are essentially utilized as primary cells, but are recharged after use rather than being discarded. Examples of these types of secondary cells primarily include portable consumer electronics and electric vehicles. Primary vs. Secondary � A Comparison The following table summarizes the pros and cons of primary and secondary batteries.
A third battery category is commonly referred to as the reserve cell. What differentiates the reserve cell from primary and secondary cells in the fact that a key component of the cell is separated from the remaining components, until just prior to activation. The component most often isolated is the electrolyte. This battery structure is commonly observed in thermal batteries, whereby the electrolyte remains inactive in a solid state until the melting point of the electrolyte is reached, allowing for ionic conduction, thus activating the battery. Reserve batteries effectively eliminate the possibility of self-discharge and minimize chemical deterioration. Most reserve batteries are used only once and then discarded. Reserve batteries are used in timing, temperature and pressure sensitive detonation devices in missiles, torpedoes, and other weapon systems. Reserve cells are typically classified into the following 4 categories.
The fuel cell represents the fourth category of batteries. Fuel cells are similar to batteries except for the fact that that all active materials are not an integral part of the device (as in a battery). In fuel cells, active materials are fed into batteries from an outside source. The fuel cell differs from a battery in that it possesses the capability to produce electrical energy as long as active materials are fed to the electrodes, but stop operating in the absence of such materials. A well-known application of fuel cells has been in cryogenic fuels used in space vehicles. Use of fuel cell technology for terrestrial applications has been slow to develop, although recent advances have generated a revitalized interest in a variety of systems with applications such as utility power, load-leveling, on-site generators and electric vehicles. |
Dry Cell Battery
Dry Cell Battery   A dry cell battery's electrolytes are contained in a low-moisture paste (represented in brown). What is a Dry Cell Battery? A dry-cell battery uses an immobilized electrolyte that minimizes moisture and allows for superior portability. The dry cell battery is one of the most commonly used types, including AA, 9-volt, and watch batteries. Dry cell batteries are different from wet cells because their electrolytes are contained in a low-moisture paste, while a wet cell has electrolytes contained in a liquid, hence the difference in names. A chemical reaction within the battery creates an electrical charge that flows from inside to an outer circuit that is connected to an electrical device. What It Is Made Of Dry cell batteries, regardless of their size, typically have the same basic components. At the center of each is a rod called a cathode, which is often made of carbon and surrounded by an electrolyte paste. Different chemicals can be used to create this paste, such as ammonium chloride and manganese dioxide, depending on the type of battery. The cathode and electrolyte paste are wrapped in paper or cardboard and sealed into a metal cylinder called an anode, which is typically made of zinc. How It Works The anode in the dry cell battery has two terminals, one that is positive and one that is negative. When a load is connected to the battery's terminals, a chemical reaction occurs between the anode and the paste that produces roughly 1.5 volts of electricity. A pin or "collector" in the middle of the battery conducts this charge out of the battery to an external circuit. This circuit physically connects to the electronic device the battery is in, providing the charge necessary for the device to function. Each set of anode, electrolyte, and cathode acts as a single cell, and multiple cells can be connected together within one dry cell battery to produce a higher overall voltage. After the load has been connected for a long time, the battery's chemicals break down and no longer produce a charge. Primary batteries should be discarded once they reach this point, while secondary batteries can be recharged through special devices. This effectively reverses the chemical reaction within each cell, allowing the battery to continue working. Defining a Dry Cell In electricity, a battery is a device consisting of one or more electrochemical cells that convert stored chemical energy into electrical energy. The dry cell is one of many general types of electrochemical cells. A dry cell has the electrolyte immobilized as a paste, with only enough moisture in it to allow current to flow. Unlike a wet cell, a dry cell can operate in any orientation without spilling, as it contains no free liquid. This versatility makes it suitable for portable equipment. By comparison, the first wet-cell batteries were typically fragile glass containers with lead rods hanging from an open top. They, therefore, needed careful handling to avoid spillage. The development of the dry-cell battery allowed for a major advance in battery safety and portability. A common dry-cell battery is the zinc-carbon battery, which uses a cell that is sometimes called the Leclanch� cell. The cell is made up of an outer zinc container, which acts as the anode. The cathode is a central carbon rod, surrounded by a mixture of carbon and manganese(IV) oxide (MnO2). The electrolyte is a paste of ammonium chloride (NH4Cl). A fibrous fabric separates the two electrodes, and a brass pin in the center of the cell conducts electricity to the outside circuit. Chemical reactions occur in every part of the battery to allow for energy storage; the reactions can be described using balanced chemical equations that delineate the electron flow. The paste of ammonium chloride reacts according to the following half-reaction: 2NH4(aq)+2e-?2NH3(g)+H2(g) The manganese(IV) oxide in the cell removes the hydrogen produced by the ammonium chloride, according to the following reaction: 2MnO2(s)+H2(g)?Mn2O3(s)+H2O(l) The combined result of these two reactions takes place at the cathode. Adding these two reactions together, we get: 2NH4(aq)+2MnO2(s)+2e?Mn2O3(s)+2NH3(g)+H2O(l) Finally, the anode half-reaction is as follows: Zn(s)?Zn2++2e- Therefore, the overall equation for the cell is: Zn(s)+2MnO2(s)+2NH4(aq)?Mn2O3(s)+H2O(l)+Zn2+2NH3(g) The potential for the above reaction is 1.50 V. Another example of a dry-cell battery is the alkaline battery. Alkaline batteries are almost the same as zinc-carbon batteries, except that the electrolyte used is potassium hydroxide (KOH) rather than ammonium chloride. In some more modern types of so-called "high-power" batteries that have a much lower capacity than standard alkaline batteries, the ammonium chloride is replaced by zinc chloride. Different Types Alkaline batteries are more popular than their older counterparts because they corrode more slowly and thus produce a charge longer. A less commonly used type of dry cell battery uses silver for the cathode rod. Nickel/Cadmium (NiCd), Nickel Metal Hydride (NiMH), and Lithium-Ion (Li-Ion) batteries are rechargeable, making them popular for use in power-hungry digital cameras and other portable devices. Proper Handling and Disposal Batteries frequently contain chemicals that are harmful if released into the environment, and should be recycled properly. Many municipal recycling programs accept batteries, though modern alkaline ones can typically be thrown away with household trash. Consumers should also consider using rechargeable batteries because they can be reused many times and can also be recycled after they no longer hold a charge. Batteries By Voltage 1.5V Batteries Specifications Description Reviews Q&A Downloads Accessories Specifications
How many mAh? A. Customer Service: The Duracell Duralock AA batteries have a capacity of 2850mAh. What is the mAh rating? A. Customer Service: The mAh rating is the capacity of the battery which measures the runtime. Duracell is the world's #1 brand for batteries! These Duralock batteries deliver dependable, long-lasting power for your household devices. Duracell AA batteries are frequently used in toys, clocks, radios, remote controls, flashlights, smoke alarms, and many more household items. Best of all, Duracell's Duralock Technology employs the cleanest chemistry to create incredibly efficient batteries, ensuring up to 10 years of power in storage and 10 years of trust. Features: Uncarded Bulk Packaging Duracell Duralock Technology features the added Duralock silver ring which ensures that when your batteries are kept in your kitchen drawer or cabinet, these batteries will still work like new when they are needed most. Whether you need to put fresh batteries in your flashlights, remote control, or emergency radio prior to the approaching storm, Duracell Duralock technology will provide you the ultimate protection you demand. CopperTop is best suited when you need reliable, long lasting life from your everyday devices! The cells are carefully packaged from bulk and are also known as: LR6, R6, MN1500, UM3, HP7 # 1500-AA Mercury-free Made in ______ | |||||||||||||||||||||||||
 Battery Size: AAA Battery Capacity: 1150mAh Battery Chemistry: Alkaline Primary Battery Voltage: 1.5 Battery Terminal Type: Button Top Battery Shelf Life: 6-8 Years Packaging: Bulk Brand: Duracell Warranty: Battery Junction 30-Day Warranty Return Policy: Battery Junction Return Policy Product Weight: 0.4oz MPN: DURACELL-AAA-MN2400-DURALOCK ______ of Origin: ___ | |||||||||||||||||||||||||
 Battery Size: C Battery Capacity: 7000mAh Battery Chemistry: Alkaline Primary Battery Voltage: 1.5 Battery Terminal Type: Button Top Battery Shelf Life: 6-8 Years Packaging: Bulk Brand: Duracell Warranty: Battery Junction 30-Day Warranty Return Policy: Battery Junction Return Policy Product Weight: 2.4oz _____ of Origin: __ | |||||||||||||||||||||||||
 Battery Size: D Battery Capacity: 15000mAh Battery Chemistry: Alkaline Primary Battery Voltage: 1.5 Battery Terminal Type: Button Top Battery Shelf Life: 6-8 Years Packaging: Bulk Brand: Duracell Warranty: Battery Junction 30-Day Warranty Return Policy: Battery Junction Return Policy Product Weight: 4.9oz ______ of Origin: ____ | |||||||||||||||||||||||||
 Battery Size: 9V Battery Capacity: 550mAh Battery Chemistry: Alkaline Primary Battery Voltage: 9 Battery Terminal Type: Snap Connector Battery Shelf Life: 6-8 Years Packaging: Bulk Brand: Duracell Warranty: Battery Junction 30-Day Warranty Return Policy: Battery Junction Return Policy Product Weight: 1.6oz Country of Origin: US |
Vehicle Battery
| Aircraft Batteries |
| Automotive Batteries |
|
How are batteries rated? What are common battery sizes? |
Non-vehicle batteries
|
Laptop Batteries How long will my fully charged battery last? |
|
Cell Phone Batteries Camera Batteries Torch Batteries Radio Batteries Two Way Radio Batteries Clock Batteries Remote Control Batteries |
| Primary Batteries (disposable) Secondary Batteries (rechargable) |
Measuring the Capacity of a Battery
| Ampere-hour |
| Converting from Watt-hours (Wh) to Amp-hours (Ah) |
| Power Analyzer |
|
The ampere-hour is frequently used in measurements of electrochemical systems such as electroplating and electrical batteries. The commonly seen milliampere-hour (mA�h or mAh) is one-thousandth of an ampere-hour (3.6 coulombs).
Batteries developing high resistance move into double-digit readings caused by any or all of the following conditions: 1. Low electrolyte level (See BU-804C: Water Loss, Acid Stratification and Surface Charge) 2. Stratification of electrolyte (See BU-804C: Water Loss, Acid Stratification and Surface Charge) 3. Sulfation of electrodes (See BU-804b: Sulfation and How to Prevent it) 4. Bad or deteriorated weld connections of the collector plates and posts 5. Collector plate cracking corroded (See BU-804a: Corrosion, Shedding and internal Short) 6. Poor battery connection at the clamps or internal to the battery |
Glossary of Battery Terms
|
Accumulator - A rechargeable battery or cell (see also Secondary battery). Ampere or Amp - An Ampere or an Amp is a unit of measurement for an electrical current. One amp is the amount of current produced by an electromotive force of one volt acting through the resistance of one ohm. Named for the French physicist Andre Marie Ampere. The abbreviation for Amp is A but its mathematical symbol is "I". Small currents are measured in milli-Amps or thousandths of an Amp. Amp Hour or Ampere-Hour - A unit of measurement of a battery's electrical storage capacity. Current multiplied by time in hours equals ampere-hours. One amp hour is equal to a current of one ampere flowing for one hour. Also, 1 amp hour is equal to 1,000 mAh Ampere-Hour Capacity - The number of ampere-hours which can be delivered by a battery on a single discharge. Anode - During discharge, the negative electrode of the cell is the anode. During charge, that reverses and the positive electrode of the cell is the anode. The anode gives up electrons to the load circuit and dissolves into the electrolyte. Aqueous Batteries - Batteries with water-based electrolytes. The electrolyte may not appear to be liquid since it can be absorbed by the battery�s separator. Actual Capacity or Available Capacity - The total battery capacity, usually expressed in ampere-hours or milliampere-hours, available to perform work. The actual capacity of a particular battery is determined by a number of factors, including the cut-off voltage, discharge rate, temperature, method of charge and the age and life history of the battery. Battery - An electrochemical device used to store energy. The term is usually applied to a group of two or more electric cells connected together electrically. In common usage, the term �battery� is also applied to a single cell, such as a AA battery. Battery Capacity - The electric output of a cell or battery on a service test delivered before the cell reaches a specified final electrical condition and may be expressed in ampere-hours, watt- hours, or similar units. The capacity in watt-hours is equal to the capacity in ampere-hours multiplied by the battery voltage. Battery Charger - A device capable of supplying electrical energy to a battery. Battery-Charge Rate - The current expressed in amperes (A) or milli amps (mA) at which a battery is charged. Cutoff Voltage, final - The prescribed lower-limit voltage at which battery discharge is considered complete. The cutoff or final voltage is usually chosen so that the maximum useful capacity of the battery is realized. The cutoff voltage varies with the type of battery and the kind of service in which the battery is used. When testing the capacity of a NiMH or NiCD battery a cutoff voltage of 1.0 V is normally used. 0.9V is normally used as the cutoff voltage of an alkaline cell. A device that is designed with too high a cutoff voltage may stop operating while the battery still has significant capacity remaining. C - Used to signify a charge or discharge rate equal to the capacity of a battery divided by 1 hour. Thus C for a 1600 mAh battery would be 1.6 A, C/5 for the same battery would be 320 mA and C/10 would be 160 mA. Because C is dependent on the capacity of a battery the C rate for batteries of different capacities must also be different. Capacity - The capacity of a battery is a measure of the amount of energy that it can deliver in a single discharge. Battery capacity is normally listed as amp-hours (or milli amp-hours) or as watt-hours. Cathode - Is an electrode that, in effect, oxidizes the anode or absorbs the electrons. During discharge, the positive electrode of a voltaic cell is the cathode. When charging, that reverses and the negative electrode of the cell is the cathode. Cell - An electrochemical device, composed of positive and negative plates and electrolyte, which is capable of storing electrical energy. It is the basic �building block� of a battery. Charge - The conversion of electric energy, provided in the form of a current, into chemical energy within the cell or battery. Charge Rate - The amount of current applied to battery during the charging process. This rate is commonly expressed as a fraction of the capacity of the battery. For example, the C/2 or C/5. Charging - The process of supplying electrical energy for conversion to stored chemical energy. Constant-Current Charge - A charging process in which the current applied to the battery is maintained at a constant value. Constant-Voltage Charge - A charging process in which the voltage applied to a battery is held at a constant value. Cycle - One sequence of charge and discharge. Deep Cycle - A cycle in which the discharge is continued until the battery reaches it�s cut-off voltage, usually 80% of discharge. Shallow Cycling - Charge and discharge cycles which do not allow the battery to approach it�s cutoff voltage. Shallow cycling of NiCd cells lead to �memory effect�. Shallow cycling is not detrimental to NiMH cells and it is the most beneficial for lead acid batteries. Cycle Life - For rechargeable batteries, the total number of charge/discharge cycles the cell can sustain before it�s capacity is significantly reduced. End of life is usually considered to be reached when the cell or battery delivers only 80% of rated ampere- hour capacity. NiMH batteries typically have a cycle life of 500 cycles, NiCd batteries can have a cycle life of over 1,000 cycles. The cycle of a battery is greatly influenced by the type depth of the cycle (deep or shallow) and the method of recharging. Improper charge cycle cutoff can greatly reduce the cycle life of a battery. Direct Current (DC) - The type of electrical current that a battery can supply. One terminal is always positive and another is always negative. Discharge - The conversion of the chemical energy of the battery into electric energy. Depth of Discharge - The amount of energy that has been removed from a battery (or battery pack). Usually expressed as a percentage of the total capacity of the battery. For example, 50% depth of discharge means that half of the energy in the battery has been used. 80% DOD means that eighty percent of the energy has been discharged, so the battery now holds only 20% of its full charge. Discharge, deep - Withdrawal of all electrical energy to the end-point voltage before the cell or battery is recharged. Discharge, high-rate - Withdrawal of large currents for short intervals of time, usually at a rate that would completely discharge a cell or battery in less than one hour. Discharge, low-rate - Withdrawal of small currents for long periods of time, usually longer than one hour. Drain - Withdrawal of current from a cell. Dry Cell - A primary cell in which the electrolyte is absorbed in a porous medium, or is otherwise restrained from flowing. Common practice limits the term �dry cell� to the Leclanch� cell, which is the common commercial type. Electrochemical Couple - The system of active materials within a cell that provides electrical energy storage through an electrochemical reaction. Electrode - An electrical conductor through which an electric current enters or leaves a conducting medium, whether it be an electrolytic solution, solid, molten mass, gas, or vacuum. For electrolytic solutions, many solids, and molten masses, an electrode is an electrical conductor at the surface of which a change occurs from conduction by electrons to conduction by ions. For gases and vacuum, the electrodes merely serve to conduct electricity to and from the medium. Electrolyte - A chemical compound which, when fused or dissolved in certain solvents, usually water, will conduct an electric current. All electrolytes in the fused state or in solution give rise to ions which conduct the electric current. Electropositivity - The degree to which an element in a galvanic cell will function as the positive element of the cell. An element with a large electropositivity will oxidize faster than an element with a smaller electropositivity. End-of-Discharge Voltage - The voltage of the battery at termination of a discharge. Energy - Output Capability - expressed as capacity times voltage, or watt-hours. Energy Density - Ratio of cell energy to weight or volume (watt-hours per pound, or watt-hours per cubic inch). Final Voltage (see Cutoff voltage) Float Charging - Method of recharging in which a secondary cell is continuously connected to a constant-voltage supply that maintains the cell in fully charged condition. Typically applied to lead acid batteries. Galvanic Cell - A combination of electrodes, separated by electrolyte, that is capable of producing electrical energy by electrochemical action. Gassing - The evolution of gas from one or both of the electrodes in a cell. Gassing commonly results from self-discharge or from the electrolysis of water in the electrolyte during charging. Internal Resistance - The resistance to the flow of an electric current within the cell or battery. Memory Effect - A phenomenon in which a cell, operated in successive cycles to less than full, depth of discharge, temporarily loses the remainder of its capacity at normal voltage levels (usually applies only to Ni-Cd cells). Note, memory effect can be induced in NiCd cells even if the level of discharge is not the same during each cycle. Memory effect is reversable. Negative Terminal - The terminal of a battery from which electrons flow in the external circuit when the cell discharges. See Positive Terminal. Nonaqueous Batteries - Cells that do not contain water, such as those with molten salts or organic electrolytes. Ohm�s Law - The formula that describes the amount of current flowing through a circuit. Ohm's Law - In a given electrical circuit, the amount of current in amperes (I) is equal to the pressure in volts (V) divided by the resistance, in ohms (R). Ohm's law can be shown by three different formulas: �To find Current I = V/R �To find Voltage V = I x R �To find Resistance R = V/I Open Circuit - Condition of a battery which is neither on charge nor on discharge (i.e., disconnected from a circuit). Open-Circuit Voltage - The difference in potential between the terminals of a cell when the circuit is open (i.e., a no-load condition). Oxidation - A chemical reaction that results in the release of electrons by an electrode�s active material. Parallel Connection - The arrangement of cells in a battery made by connecting all positive terminals together and all negative terminals together. The voltage of the group remains the same as the voltage of the individual cell. The capacity is increased in proportion to the number of cells. Polarity - Refers to the charges residing at the terminals of a battery. Positive Terminal - The terminal of a battery toward which electrons flow through the external circuit when the cell discharges. See Negative Terminal. Primary Battery - A battery made up of primary cells. See Primary Cell. Primary Cell - A cell designed to produce electric current through an electrochemical reaction that is not efficiently reversible. The cell, when discharged, cannot be efficiently recharged by an electric current. Alakline, lithium, and zinc air are common types of primary cells. Rated Capacity - The number of ampere-hours a cell can deliver under specific conditions (rate of discharge, end voltage, temperature); usually the manufacturer�s rating. Rechargeable - Capable of being recharged; refers to secondary cells or batteries. Recombination - State in which the gases normally formed within the battery cell during its operation, are recombined to form water. Reduction - A chemical process that results in the acceptance of electrons by an electrode�s active material. Seal - The structural part of a galvanic cell that restricts the escape of solvent or electrolyte from the cell and limits the ingress of air into the cell (the air may dry out the electrolyte or interfere with the chemical reactions). Secondary Battery - A battery made up of secondary cells. See Storage Battery; Storage Cell. Self Discharge - Discharge that takes place while the battery is in an open-circuit condition. Separator - The permeable membrane that allows the passage of ions, but prevents electrical contact between the anode and the cathode. Series Connection - The arrangement of cells in a battery configured by connecting the positive terminal of each successive cell to the negative terminal of the next adjacent cell so that their voltages are cumulative. See Parallel Connection. Shelf Life - For a dry cell, the period of time (measured from date of manufacture), at a storage temperature of 21 degrees C (69 degrees F), after which the cell retains a specified percentage (usually 90%) of its original energy content. Short-Ciruit - A condition that occurs when a short electrical path is unintentionally created. Batteries can supply hundreds of amps if short-circuited, potentially melting the terminals and creating sparks. Short-Circuit Current - That current delivered when a cell is short-circuited (i.e., the positive and negative terminals are directly connected with a low-resistance conductor). Starting-Lighting-Ignition (SLI) Battery - A battery designed to start internal combustion engines and to power the electrical systems in automobiles when the engine is not running. SLI batteries can be used in emergency lighting situations. Stationary Battery - A secondary battery designed for use in a fixed location. Storage Battery - An assembly of identical cells in which the electrochemical action is reversible so that the battery may be recharged by passing a current through the cells in the opposite direction to that of discharge. While many non-storage batteries have a reversible process, only those that are economically rechargeable are classified as storage batteries. Synonym: Accumulator; Secondary Battery. See Secondary Cell. Storage Cell - An electrolytic cell for the generation of electric energy in which the cell after being discharged may be restored to a charged condition by an electric current flowing in a direction opposite the flow of current when the cell discharges. Synonym: Secondary Cell. See Storage Battery. Taper Charge - A charge regime delivering moderately high-rate charging current when the battery is at a low state of charge and tapering the current to lower rates as the battery becomes more fully charged. Terminals - The parts of a battery to which the external electric circuit is connected. Thermal Runaway - A condition whereby a cell on charge or discharge will destroy itself through internal heat generation caused by high overcharge or high rate of discharge or other abusive conditions. Trickle Charging - A method of recharging in which a secondary cell is either continuously or intermittently connected to a constant-current supply that maintains the cell in fully charged condition. Vent - A normally sealed mechanism that allows for the controlled escape of gases from within a cell. Volt - The unit of measurement of electromotive force, or difference of potential, which will cause a current of one ampere to flow through a resistance of one ohm. Named for Italian physicist Alessandro Volta (1745-1827). Voltage, cutoff - Voltage at the end of useful discharge. (See Voltage, end-point.) Voltage, end-point - Cell voltage below which the connected equipment will not operate or below which operation is not recommended. Voltage, nominal - Voltage of a fully charged cell when delivering rated current. Watt - A measurement of total power. It is amperes multiplied by volts. 120 volt @ 1 amp = 12 volts @ 10 amps. Wet Cell - A cell, the electrolyte of which is in liquid form and free to flow and move. |
Wet Cell, Gel-Cell and Absorbed Glass Mat (AGM)
|
What is a wet-cell battery and how does it differ from a dry-cell battery? A wet-cell battery is the original type of rechargeable battery. It is commonly found in aviation, electric utilities, energy storage and cellphone towers. The battery contains a liquid electrolyte such as sulfuric acid, a dangerous corrosive liquid that damages what it comes into contact with. A dry-cell battery does not contain liquid. Smaller dry-cell batteries, such as alkaline or lithium ion, are typically used in portable electronics, such as toys, phones and laptops. - |
Batteries for Portable Devices
Portable electronic devices, containing batteries
|
Cell phones, laptop, camera, smart phones, PDAs, games, tablets, watches, etc.
Most consumer personal electronic devices containing batteries are allowed in carry-on and checked baggage, including but not limited to cell phones, smart phones, PDAs, electronic games, tablets, laptop computers, cameras, camcorders, watches, calculators, etc. This covers typical dry cell batteries and lithium metal and lithium ion batteries for consumer electronics (AA, AAA, C, D, button cell, camera batteries, laptop batteries, etc.) Devices containing lithium metal or lithium ion batteries (laptops, smartphones, tablets, etc.) should be carried in carry-on baggage when possible. When these devices must be carried in checked baggage, they should be turned completely off, protected from accidental activation, and packed so they are protected from damage. Spare (uninstalled) lithium metal and lithium ion batteries are always prohibited in checked baggage and must be placed in carry-on. When a carry-on bag is checked at the gate or at planeside, any spare lithium batteries must be removed from the bag and kept with the passenger in the aircraft cabin. See separate entry in this chart for electronic cigarettes and vaping devices. These are always prohibited in checked baggage. What Battery Brand Provides Power the Longest? 1-Sunbeam 2-Energizer 3-Duracell 4-Rayovac Sunbeam Super Heavy Duty AA Batteries, 8-ct. Packs 
Images for Sunbeam AA batteries Super heavy duty mercury-free "AA" batteries are ideal for low-drain devices. Specifications Case Quantity: 48 Assortment Details: 8-ct. packs of Sunbeam® super heavy duty "AA" batteries. Lithium-ion battery A lithium-ion battery or Li-ion battery (abbreviated as LIB) is a type of rechargeable battery in which lithium ions move from the negative electrode to the positive electrode during discharge and back when charging. Li-ion batteries use an intercalated lithium compound as one electrode material, compared to the metallic lithium used in a non-rechargeable lithium battery. The electrolyte, which allows for ionic movement, and the two electrodes are the constituent components of a lithium-ion battery cell. How Does a Lithium-ion Battery Work? Lithium-ion batteries power the lives of millions of people each day. From laptops and cell phones to hybrids and electric cars, this technology is growing in popularity due to its light weight, high energy density, and ability to recharge. So how does it work? This animation walks you through the process. The Basics A battery is made up of an anode, cathode, separator, electrolyte, and two current collectors (positive and negative). The anode and cathode store the lithium. The electrolyte carries positively charged lithium ions from the anode to the cathode and vice versa through the separator. The movement of the lithium ions creates free electrons in the anode which creates a charge at the positive current collector. The electrical current then flows from the current collector through a device being powered (cell phone, computer, etc.) to the negative current collector. The separator blocks the flow of electrons inside the battery. Charge/Discharge While the battery is discharging and providing an electric current, the anode releases lithium ions to the cathode, generating a flow of electrons from one side to the other. When plugging in the device, the opposite happens: Lithium ions are released by the cathode and received by the anode. Energy Density vs. Power Density The two most common concepts associated with batteries are energy density and power density. Energy density is measured in watt-hours per kilogram (Wh/kg) and is the amount of energy the battery can store with respect to its mass. Power density is measured in watts per kilogram (W/kg) and is the amount of power that can be generated by the battery with respect to its mass. To draw a clearer picture, think of draining a pool. Energy density is similar to the size of the pool, while power density is comparable to draining the pool as quickly as possible. The Vehicle Technologies Office works on increasing the energy density of batteries, while reducing the cost, and maintaining an acceptable power density. Electric Drive Systems Research and Development Battery electric vehicle 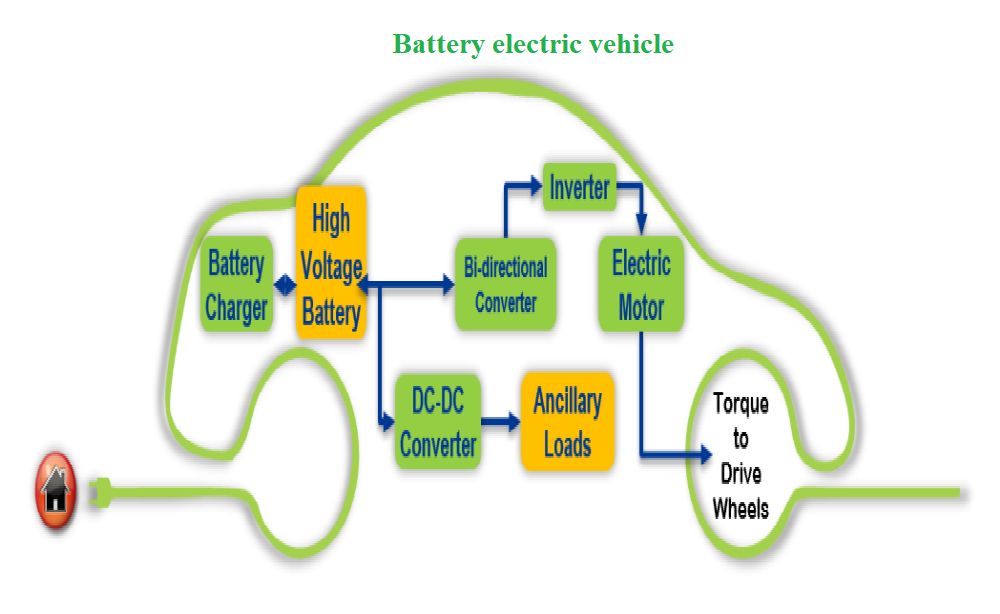
Electric drive technologies, including the electric motor, inverter, boost converter, and on-board charger, are essential components of hybrid and plug-in electric vehicles (PEV) propulsion systems. |
