Coronary Circulation
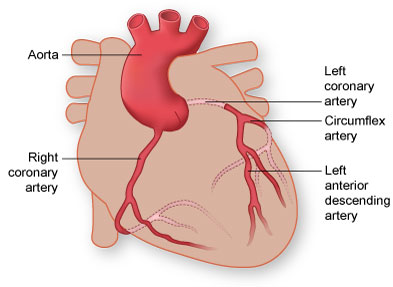
The heart muscle, like every other organ or tissue in your body, needs oxygen-rich blood to survive. Blood is supplied to the heart by its own vascular system, called coronary circulation.
The aorta (the main blood supplier to the body) branches off into two main coronary blood vessels (also called arteries). These coronary arteries branch off into smaller arteries, which supply oxygen-rich blood to the entire heart muscle.
The right coronary artery supplies blood mainly to the right side of the heart. The right side of the heart is smaller because it pumps blood only to the lungs.
The left coronary artery, which branches into the left anterior descending artery and the circumflex artery, supplies blood to the left side of the heart. The left side of the heart is larger and more muscular because it pumps blood to the rest of the body.
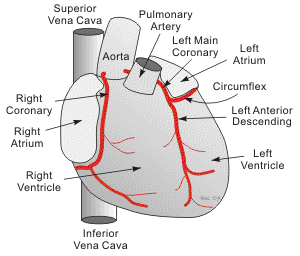
The major vessels of the coronary circulation are the left main coronary that
divides into left anterior descending and circumflex branches, and the right main coronary
artery. The left and right coronary arteries originate at the base of the aorta
from openings called the coronary ostia located behind the aortic valve
leaflets.
The left and right coronary arteries and their branches lie on the surface of
the heart, and therefore are sometimes referred to as the epicardial coronary
vessels. These vessels distribute blood flow to different regions of the
heart muscle. When the vessels are not diseased, they have a low
vascular resistance relative to their
more distal and smaller branches that comprise the
microvascular network. As in all
vascular beds, it is the small arteries and arterioles in the microcirculation
that are the primary sites of vascular resistance, and therefore the primary
site for regulation of blood flow. The arterioles branch into numerous
capillaries that lie adjacent to the cardiac myocytes. A high
capillary-to-cardiomyocyte ratio and short diffusion distances ensure adequate
oxygen delivery to the myocytes and removal of metabolic waste products from the
cells (e.g., CO2 and H+). Capillary blood flow enters
venules that join together to form cardiac veins that drain into the coronary
sinus located on the posterior side of the heart, which drains into the
right atrium. There are also anterior cardiac veins and thesbesian
veins drain directly into the cardiac chambers.
Although there is considerable heterogeneity among people, the following
table indicates the regions of the heart that are generally supplied by the
different coronary arteries. This anatomic distribution is important because
these cardiac regions are assessed by 12-lead
ECGs to help localize ischemic or infarcted regions, which can be loosely
correlated with specific coronary vessels; however, because of vessel
heterogeneity, actual vessel involvement in ischemic conditions needs to be
verified by coronary angiograms or other imaging techniques.
| Anatomic Region of Heart |
Coronary Artery (most likely associated) |
| Inferior |
Right coronary |
| Anteroseptal |
Left anterior descending |
| Anteroapical |
Left anterior descending (distal) |
| Anterolateral |
Circumflex |
| Posterior |
Right coronary artery |
The following summarizes important features of coronary blood flow:
Flow is tightly coupled to oxygen demand. This is
necessary because the heart has a very high basal oxygen
consumption (8-10 ml O2/min/100g) and the highest A-VO2
difference of a major organ (10-13 ml/100 ml). In non-diseased coronary
vessels, whenever cardiac activity and oxygen consumption increases, there is an increase
in coronary blood flow (active hyperemia) that is nearly
proportionate to the increase in oxygen consumption.
Good autoregulation between
60 and 200 mmHg perfusion pressure helps to maintain normal coronary blood flow whenever
coronary perfusion pressure changes due to changes in aortic pressure.
Adenosine is an important
mediator of active hyperemia and autoregulation. It serves as a metabolic coupler
between oxygen consumption and coronary blood flow. Nitric oxide
is also an important regulator of coronary blood flow.
Activation of sympathetic nerves innervating
the coronary vasculature causes only transient vasoconstriction mediated by 1-adrenoceptors.
This brief (and small) vasoconstrictor response is followed by vasodilation
caused by enhanced production of vasodilator metabolites
(active hyperemia) due to increased mechanical and
metabolic activity of the heart resulting from β1-adrenoceptor
activation of the myocardium. Therefore, sympathetic activation to the
heart results in coronary vasodilation and increased coronary flow due to increased
metabolic activity (increased heart rate, contractility) despite direct
vasoconstrictor effects of
sympathetic activation on the coronaries. This is termed
"functional sympatholysis."
Parasympathetic stimulation of the
heart (i.e., vagal nerve activation) elicits modest coronary vasodilation (due to the
direct effects of released acetylcholine on the coronaries). However, if parasympathetic
activation of the heart results in a significant decrease in myocardial
oxygen demand due to a reduction in heart rate, then intrinsic metabolic mechanisms will increase coronary vascular
resistance by constricting the vessels.
Progressive ischemic coronary artery disease results in
the growth of new vessels (termed angiogenesis) and collateralization within the
myocardium. Collateralization increases myocardial blood supply by increasing the number
of parallel vessels, thereby reducing vascular resistance within the myocardium.
(shown to the right) during
systole markedly affects coronary flow; therefore, most of the coronary flow occurs during
diastole. Because of extravascular compression, the endocardium is more susceptible to ischemia especially at lower perfusion pressures. Furthermore, with
tachycardia there is relatively less time available for coronary flow during diastole to
occur – this is particularly significant in patients with coronary artery disease
where coronary flow reserve (maximal flow capacity) is reduced.
In the presence of coronary artery disease,
coronary blood flow may be reduced. This will increase
oxygen extraction from the coronary blood and
decrease the venous oxygen content. This leads to tissue
hypoxia and angina.
If the lack of blood flow is due to a fixed stenotic lesion in the coronary
artery (because of atherosclerosis), blood flow can be improved within that
vessel by 1) placing a stent within the vessel to expand the lumen, 2) using an
intracoronary angioplasty balloon to stretch the vessel open, or 3) bypassing
the diseased vessel with a vascular graft. If the insufficient blood flow is
caused by a blood clot (thrombosis), a thrombolytic drug that dissolves clots
may be administered. Anti-platelet drugs and aspirin are commonly used to
prevent the reoccurrence of clots. If the reduced flow is due to coronary
vasospasm, then coronary vasodilators can be given
(e.g.,
nitrodilators,
calcium-channel blockers) to reverse and prevent vasospasm.
Where does your heart get its own oxygen? From the coronary arteries that surround the heart muscle and pump blood into every portion of it. The right coronary artery supplies blood to the right and left sides of your heart. The left main coronary artery has two main branches - the left anterior descending artery, which feeds the front left side of the heart, and the circumflex artery that feeds the back of your heart. These main coronary arteries have many branches that supply your heart muscle with oxygen-rich blood.
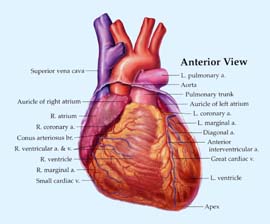
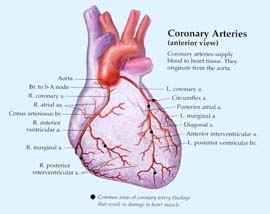
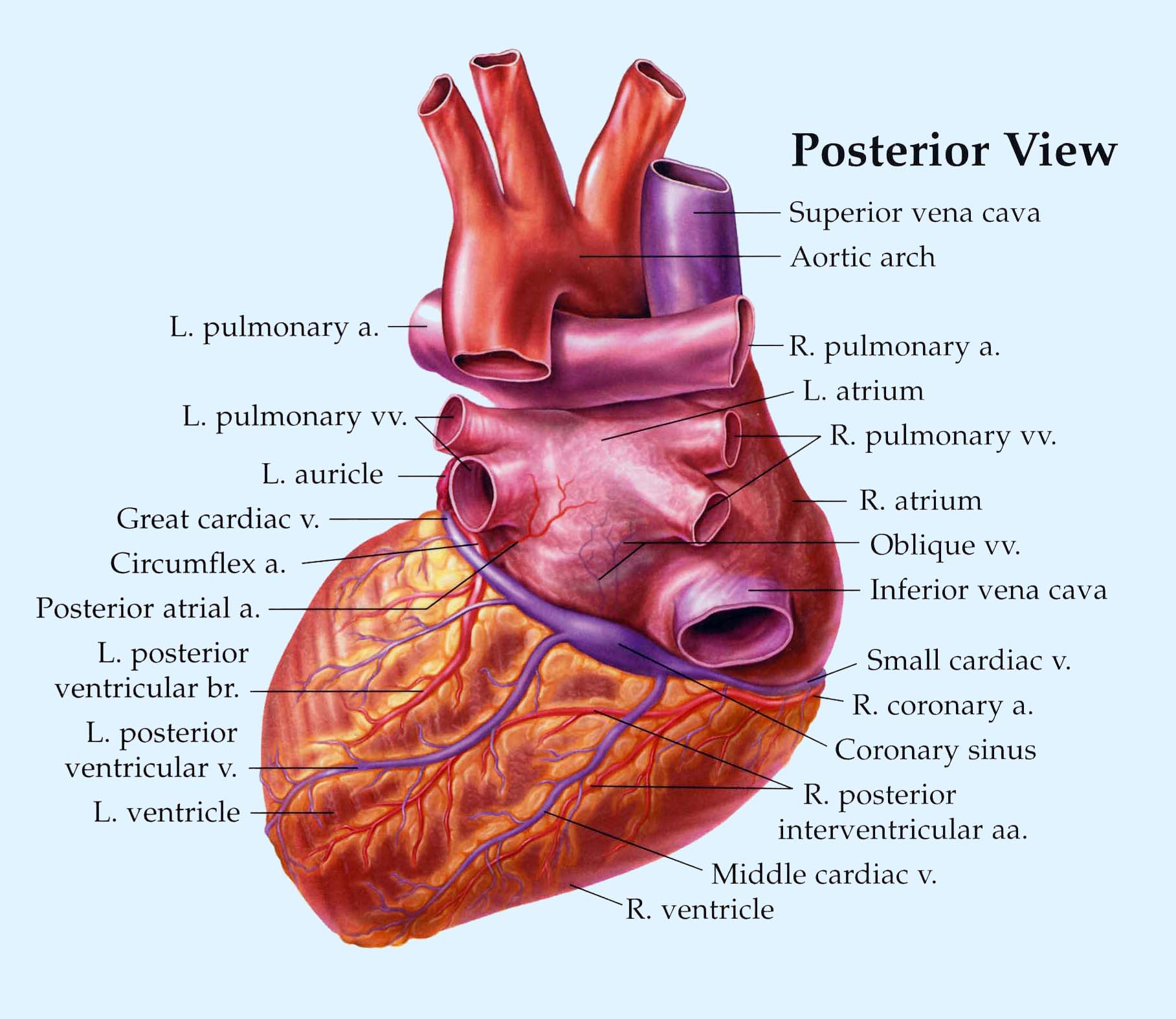
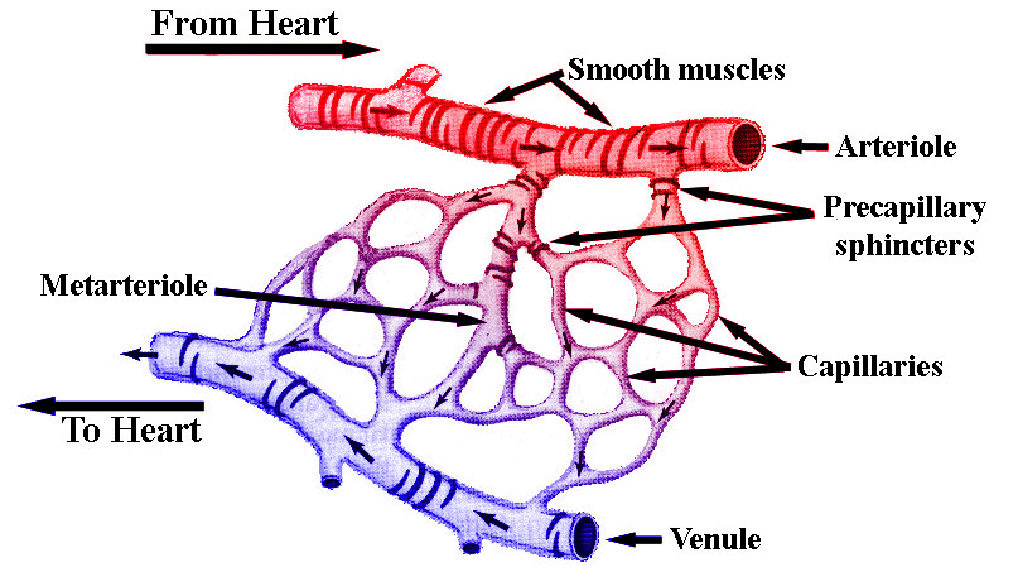
Figure 2. The microcirculation including arterioles, capillaries and venules. The capillaries lie between, or connect, the arterioles and venules. Capillaries form extensive branching networks that dramatically increase the surface areas available for the rapid exchange of molecules. A metarteriole is a vessel that emerges from an arteriole and supplies a group of 10 to 100 capillaries. Both the arteriole and the proximal portion of the metarterioles are surrounded by smooth muscle fibers whose contractions and relaxations regulate blood flow through the capillary bed. Typically, blood flows intermittently through a capillary bed due to the periodic contractions of the smooth muscles (5-10 times per minute, vasomotion), which is regulated both locally (metabolically) and by sympathetic control.
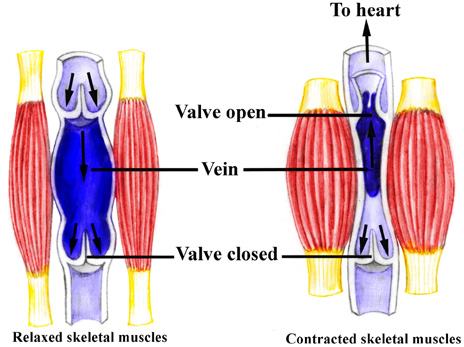
Figure 3. Contractions of the skeletal muscles aid in returning blood to the heart—skeletal muscle pump. While standing at rest, the relaxed vein acts as a reservoir for blood; contractions of limb muscles not only decrease this reservoir size (venous diameter), but also actively force the return of more blood to the heart. Note that the resulting increase in blood flow due to the contractions is only towards the heart due to the valves in the veins.
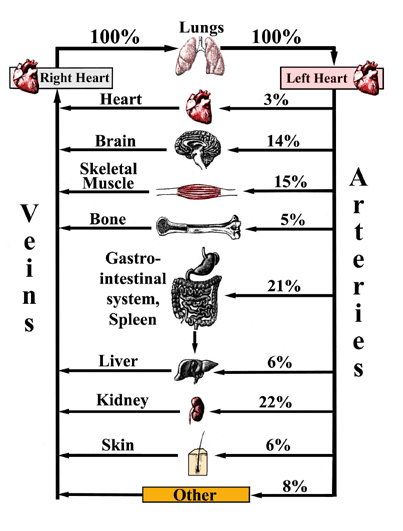
Figure 6. Provided is a functional representation of the blood circulatory system. The percentages indicate the approximate relative percentages of the cardiac output that is delivered, at a given moment in time, to the major organ systems within the body.
|








